Particulate Matter and Acid Deposition Modeling for the Southern Appalachian Mountains Initiative
James Boylan, James Wilkinson, Yueh-Jiun Yang, Talat Odman and Armistead Russell.
Georgia Institute of Technology, 200 Bobby Dodd Way, Atlanta, GA 30332-0512
ABSTRACT
As part of the Southern Appalachians Mountains Initiative (SAMI), the effects of emissions controls on air quality are being assessed using an integrated, “one‑atmosphere” modeling approach. The modeling system consists of RAMS for meteorology, EMS-95 for emissions and the Urban-to-Regional Multiscale (URM) model for transport and chemistry. With this system, the evolution of primary and secondary gas-phase and aerosol-phase pollutants, can be followed and both PM10 and PM2.5 can be simulated. EMS-95 has the ability to inventory size- and species-resolved aerosol emissions. URM, as applied to this project, includes a wet deposition module, an aerosol module, and a direct sensitivity analysis module, which simultaneously yields three-dimensional sensitivities of pollutant concentrations and deposition fluxes to emissions. Nine episodes (each seven to ten days in duration) will be simulated over the eastern United States using a fine grid of 12 km in and around the SAMI states, and successively coarser grids, up to 192 km near the boundaries of the domain. The results are used to develop seasonal and annual air quality metrics and their response to emission controls, for an assessment of the visibility and acid deposition in the region. In this paper, URM is evaluated for its ability to accurately simulate PM2.5 concentrations, as well as, the deposition of acids in four episodes. When compared to data from IMPROVE sites, the model predicted concentrations for most fine aerosol species with less than 50 percent normalized error. The wet deposition amounts are in good agreement with the NADP observations when the high spatial and temporal variability of precipitation is factored into the performance metrics. These results suggest that the modeling system can be confidently used for assessing control strategy impacts. In this study, the domain has been divided into eight source regions, and using the direct sensitivity analysis feature of URM, the relationships between the aerosol levels in the southern Appalachians and the emissions from various regions have been studied. SO2 emission reductions from different regions displayed very different levels of impact at various sites within the SAMI region.
INTRODUCTION
Studies that have been conducted in national parks and national forest wilderness areas of the southern Appalachian Mountains have documented adverse air pollution effects on visibility, streams, soil, and vegetation. Although it is known that air pollution levels which currently affect park and wilderness resources come from existing sources of pollution - large and small, mobile and stationary, near and distant, the relative contribution of each source type to the regional air pollution problem is not well quantified. The 1990 Clean Air Act Amendments (CAAA) required major reductions in airborne pollutants. Although the reductions are expected to produce air quality improvements, there is uncertainty whether the results will be enough to protect and preserve the ecosystems and natural resources of the southern Appalachians, especially in Class I areas.
The Urban-to-Regional Multiscale (URM) model5 has been applied to the SAMI modeling domain to characterize the air pollution formation processes that affect air quality in the southern Appalachian Mountains. The URM model results will be used to assess emission control strategies designed to reduce atmospheric pollutants in the Class I areas.
The URM model is a three-dimensional Eulerian photochemical model that accounts for the transport and chemical transformation of pollutants in the atmosphere. The URM model uses a finite element, variable mesh transport scheme6. The gas-phase reaction kinetics are calculated using the SAPRC chemical mechanism2. This mechanism has been updated to account for a more accurate isoprene chemistry among other reactions. URM uses variable size grids in its horizontal domain to effectively capture the details of pollution dynamics without being computationally intensive. The URM model has been expanded to include aerosol dynamics and wet deposition scavenging processes. The ISORROPIA aerosol module4, the Reactive Scavenging Module1 for acid deposition, and the Direct Decoupled Method9 for gas and aerosol sensitivities have been incorporated into the URM framework to produce an integrated, “one atmosphere” airshed model. The URM model and its setup for this project are discussed in more detail in Odman et al.7
AEROSOL MODULE
The aerosol module is capable of simulating concentrations of all major primary and secondary components of atmospheric PM. There are three groups of aerosol species that are considered in the aerosol routine. These include inert species, inorganic equilibrium species, and organic species. The inert species include magnesium, potassium, calcium, elemental carbon, and an “other PM” group which includes all other inert PM species. The inorganic equilibrium species include sulfate, nitrate, ammonium, sodium, chloride, and hydrogen ion. The organic aerosols are represented by a lumped species which is the sum of numerous condensable organics resulting from the oxidation of organic gases.
A sectional approach is used for characterization of the continuous aerosol size distribution by using four size bins: smaller than 0.156 µm, 0.156 - 0.625 µm, 0.625 - 2.5 µm, and 2.5 - 10.0 µm. The module simulates mass transfer and particle growth occurring between the gaseous and aerosol species during condensation and evaporation. The effects of nucleation and coagulation are ignored. The ISORROPIA algorithm is used to model inorganic atmospheric aerosols. ISORROPIA is a computationally efficient and rigorous thermodynamic model that predicts the physical state and composition of the sodium-ammonium-chloride-sulfate-nitrate-water aerosol system. The aerosol particles are assumed to be internally mixed, meaning that all particles of the same size have the same composition. The possible species for each phase are:
Gas Phase: NH3, HNO3, HCl, H2O
Liquid Phase: NH4+, Na+, H+, Cl-, NO3-, SO42-, HSO4-, OH-, H2O
Solid Phase: (NH4)2SO4, NH4HSO4, (NH4)3H(SO4)2, NH4NO3, NH4Cl, NaCl, NaNO3,
NaHSO4,
Na2SO4, H2SO4
The ISORROPIA mechanism contains fifteen equilibrium reactions that are solved for in conjunction with their equilibrium constants4.
The production of condensable organic species from oxidation of gaseous organic compounds is based on the organic yields reported by Pandis et al.8 The formation of condensable organic aerosol species is done in the chemistry module, followed by the distribution of condensed organic aerosols to the four size bins in the aerosol routine. Also, an algorithm to simulate particle deposition and gravitational settling for particles of various sizes has been added. Inputs to the aerosol module include temperature, relative humidity, air density, and gas and aerosol concentrations. Outputs from the module are the updated equilibrium concentrations for the gas (HNO3, NH3, and HCl) and the aerosol species.
REACTIVE SCAVENGING MODULE
The Reactive Scavenging Module (RSM) uses synoptic scale temperature and precipitation rates to simulate a field of representative clouds which are defined by scavenging rates, water profiles, and wind fields. The module simulates the time dependent chemical kinetic interaction of these clouds with the gas and aerosol species, as well as, simulating vertical convective transport within a column of air1. Scavenging processes within the module include gas, aerosol, and microphysical scavenging. Gas scavenging can occur in cloud water (equilibrium process based on species solubilities), rain water (mass transfer rates are calculated for most species), and snow (limited to nitric acid). Aerosol scavenging is treated by nucleation and by inertial impaction processes. Microphysical scavenging refers to processes that not only transfer water from one water category to another, but also transfer the water-bound pollutants.
There are a number of input parameters that are required to be passed from URM to the RSM module, including temperatures and pressures for all vertical layers at each computational node. There are five additional meteorological parameters that are required to be passed: (1) total area averaged precipitation rate of convective and stratiform clouds, (2) fraction of precipitation associated with convective rain, (3) fraction of grid area covered by convective storms, (4) cloud top height of convective storm, and (5) cloud top height of stratiform storm.
The other required inputs are the gas and aerosol concentration profiles for the species that will be scavenged and/or transported via the convective clouds. The scavenged species are sulfur dioxide, aerosol sulfate, ozone, nitric acid, aerosol nitrate, hydrogen peroxide, ammonia, ammonium aerosol, and soluble crustals (Mg and Ca). Other gas and aerosol species are passed into the RSM module where vertical convective transport is simulated. Output from the module includes updated concentration profiles for all the species affected by scavenging and convective cloud transport, in addition to wet deposition mass fluxes for SO2, SO4, NO3, H2O2, NH4, Mg, Ca, K, and H-ion.
SAMI MODELING DOMAIN
The URM modeling domain covers the eastern half of the United States. The multiscale grid dimensions correspond to 192, 96, 48, 24, and 12 km. We have placed the fine 12 km grid over the southern Appalachian Mountains and the adjacent areas that are expected to most directly influence the air quality in the region of interest. The coarse grid is placed over the boundary cells and in areas that are not expected to significantly contribute to the air quality in the southern Appalachian Mountains. The domain height is 12,867 m and is divided into seven vertical grid layers. The thickness of each layer from the ground to the top of the domain are: 19 m, 43 m, 432 m, 999 m, 1779 m, 3588 m, and 6007 m. The use of finer resolution near the surface of the domain, as compared to the coarser resolution aloft, allows the steeper concentration gradients that typically exist in the near-surface troposphere and the evolution of the mixing depths during the day to be captured. Also, the SAMI modeling domain has been divided up into sub-domains in order to perform sensitivity analysis. Figure 1 shows the URM modeling grid along with the eight sub-domains that will be considered in assessing emission reductions.
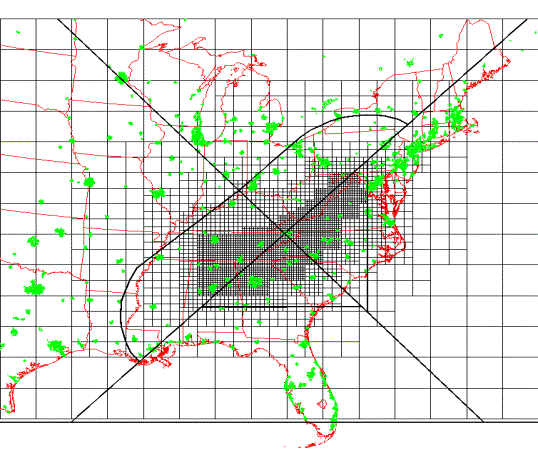
Figure 1: SAMI modeling domain and sensitivity sub-domains
MODEL PERFORMANCE
A total of nine episodes will be used to develop seasonal and annual air quality metrics to assess visibility and acid deposition problems in the region. This paper focuses on four episodes: July 11 - 19, 1995; May 24 - 29, 1995; May 11 - 17, 1993; and March 23 - 31, 1993. A comprehensive set of statistical calculations has been performed to determine the ability of the model to accurately estimate ambient aerosol concentrations and acid deposition mass fluxes. Among the statistical measures examined are mean bias, normalized bias, mean error, and normalized error. The statistical calculations were done using the Modeling Analysis and Plotting System (MAPS) package3. The results will be summarized below.
Aerosol
Performance
Aerosol performance was evaluated by comparing modeling results to observations taken from the Interagency Monitoring of Protected Visual Environments (IMPROVE ) monitoring network. The species that were compared include fine (< 2.5 mm) sulfate, fine nitrate, fine ammonium, fine elemental carbon, fine organic carbon, fine soils (crustals), total PM2.5, and total PM10. There are eighteen IMPROVE monitoring sites in the modeling domain. However, five of those sites are located near or on the boundary of the modeling domain and are easily influenced by the boundary conditions. Therefore, only observations from the remaining thirteen stations are used to determine the aerosol model performance. Table 1 lists the resolution of the URM grid cells containing these stations. IMPROVE measurements are taken twice each week (Wednesday and Saturday) and are reported as a twenty-four hour average concentration. The four nearest nodes to each IMPROVE station are distance weighted and used to determine the aerosol concentration at each monitoring site.
Table 2 shows a summary of the normalized mean error for all the stations in the SAMI states for each day that IMPROVE measurements were available throughout the four modeled episodes. The normalized mean error (NME) is calculated as:

where ![]() is the model-estimated
24-hour aerosol concentration at station i,
is the model-estimated
24-hour aerosol concentration at station i,
![]() is the observed
24-hour aerosol concentration at station i,
and N equals the number of
estimate-observation pairs drawn from all valid monitoring station data on the
simulation day of interest.
is the observed
24-hour aerosol concentration at station i,
and N equals the number of
estimate-observation pairs drawn from all valid monitoring station data on the
simulation day of interest.
Table 1: URM grid resolution for IMPROVE stations.
|
IMPROVE STATION |
Grid cell size (km) |
|
Brigantine (NJ) |
48 |
|
Dolly Sods/Otter Creek (WV) |
12 |
|
Great Smoky Mountains (TN) |
12 |
|
Jefferson/James River Face (VA) |
12 |
|
Lye Brook (VT) |
96 |
|
Mammoth Cave (KY) |
24 |
|
Okefenokee (GA) |
96 |
|
Cape Romain (SC) |
48 |
|
Shenandoah (VA) |
12 |
|
Shining Rock (NC) |
12 |
|
Sipsy (AL) |
12 |
|
Upper Buffalo (AR) |
96 |
|
Washington, D.C. |
24 |
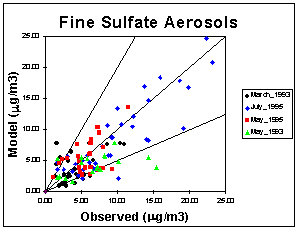
Typically, the largest portion of fine PM consists of sulfate. Sulfate is produced by the gas phase reaction of SO2 with OH or heterogeneously by reacting SO2 with H2O2 and/or ozone when a rain, cloud, or fog droplet is present. The mean normalized mean error is calculated by averaging the normalized mean errors for each of the 10 IMPROVE days that measurements were taken. These values are reported for each species in Table 2. The mean normalized mean error for sulfate is less than 45%. Figure 2 shows a scatter plot of observed versus modeled sulfate concentrations for all the IMPROVE stations in the domain for each episode. Aerosol nitrate is formed by the condensation of nitric acid into the aqueous or salt complex form. Its concentration depends on the amount of gas-phase nitric acid, ammonia, and sulfate that is available. The nitrate concentrations are usually low (less than 1 mg/m3); therefore, the normalized error can be very high. Ammonium usually presents
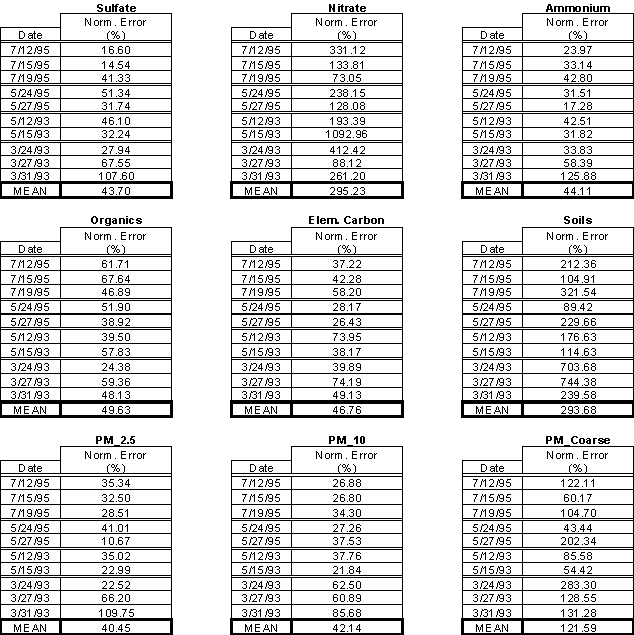
Table 2: Aerosol mean normalized errors for July '95, May '95, May '93, and
March '93 episodes.
itself in the form of ammonium sulfate ((NH4)2SO4), ammonium bisulfate (NH4HSO4), ammonium nitrate (NH4NO3), and/or mixed salts. The ammonium concentration primarily depends on the amount of sulfate and gas-phase ammonia that is available. There are no direct measurements of ammonium at the IMPROVE stations. Therefore, for the purpose of ammonium performance evaluation, it has been assumed that the sulfate and nitrate are completely neutralized with NH4. The mean normalized mean error for the ammonium aerosol is similar to that of sulfate (i.e. less than 45%).
The amount of organic aerosols formed in the atmosphere is determined by using measured organic aerosol yields. Almost all of the model predictions are biased low. This can possibly be attributed to an under prediction in the organic aerosol yields that were used or a deficiency in the emission inventory. The mean normalized mean error is still less than 50%. Elemental carbon (EC) is an inert primary emission species. The observations for EC are typically very low (around 0.5 mg/m3). The model results match well with observations and have an average mean error of less than 0.25 mg/m3. The aerosol species that are lumped into “soils” consist of calcium and “other” PM. The predictions typically are biased high. There are high uncertainties in the emission inventories for these crustal species, which may lead to the overprediction.
In order to determine PM2.5, all the aerosol species for the first 3 size bins were summed together. To determine the PM10 concentrations, the coarse aerosol fractions (2.5 – 10.0 mm) were added to the PM2.5 concentrations. There is good agreement between model predictions and observation (typically less than 40% normalized mean error) for most of the episode days, except for the March 1993 episode. This larger discrepancy in the March 1993 episode is mainly due to the high errors associated with the sulfate, ammonium, and soils.
Acid
Deposition Performance
Wet acid deposition performance was evaluated by comparing modeling results to observations taken from the National Atmospheric Deposition Program (NADP) monitoring network. The species that were compared include sulfate, nitrate, ammonium, hydrogen ion, and crustal cations (Mg and Ca). NADP measurements are taken once each week (Tuesday) and the concentrations and precipitation are reported as a 7-day cumulative. Dividing the measured concentration by the precipitation results in depositions with units of mass flux. Since there is a large spatial variation in precipitation and the RAMS model results do not match observed precipitation at exact locations, the NADP observations are compared to the best (i.e., closest to the observation) model result within a 30 km radius from the monitoring site. There are eighty-three NADP monitoring sites in the modeling domain. However, since wet deposition is very localized, only data from the fourteen stations in the 12 km grids are used to determine model performance. These stations are listed in Table 3.
Table 3: NADP stations falling into the 12-km URM grid
|
STATION |
COUNTY |
STATE |
|
Sand Mountain Exp. Station |
De Kalb |
AL |
|
Georgia Station |
Pike |
GA |
|
Lilley Cornett Woods |
Letcher |
KY |
|
Coweeta |
Macon |
NC |
|
Piedmont Research Station |
Rowan |
NC |
|
Mt. Mitchell |
Yancey |
NC |
|
Walker Branch Watershed |
Anderson |
TN |
|
Wilburn Chapel |
Giles |
TN |
|
Great Smokey Mountains National Park – Elkmont |
Sevier |
TN |
|
Charlottesville |
Albemarle |
VA |
|
Horton’s Station |
Giles |
VA |
|
Shenandoah National Park |
Madison |
VA |
|
Babcock State Park |
Fayette |
WV |
|
Parsons |
Tucker |
WV |
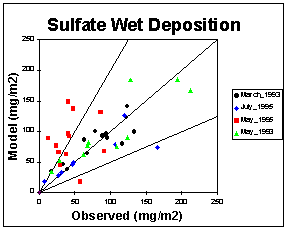
Sulfate and nitrate are two of the most important wet deposition species. They both show low normalized mean error. The average mass fluxes and normalized mean errors for the four wet deposition episodes are located in Table 4. The mean normalized mean errors for sulfate and nitrate are 35.4% and 31.8%, respectively. Figure 3 shows a scatter plot of observed versus modeled wet sulfate deposition mass fluxes for all the NADP stations in the 12 km grid for each episode. The correlation between modeled and observed sulfate mass fluxes are very good, except for the May 1995 episode which shows an overprediction.
Table 4: Wet deposition performance for July '95, May '95, May '93, and March '93 episodes.

aerosol concentrations wet deposition mass fluxes
SENSITIVITY ANALYSIS
Using air quality models to calculate the sensitivity of gas and aerosol phase concentrations and wet deposition fluxes to input and system parameters is important in determining the most effective control strategies. In order to most efficiently and accurately determine sensitivities, the Decoupled Direct Method (DDM) has been integrated into the model. Using direct derivatives of the equations governing the evolution of species concentrations, the local sensitivities to a variety of model parameters and inputs are computed simultaneously with the species concentrations.
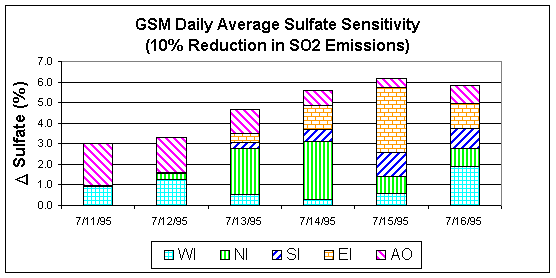
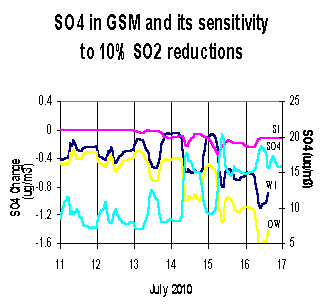
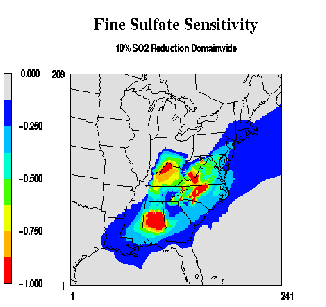
Figure 4:
Fine sulfate sensitivity to a 10 percent SO2 reduction.
Preliminary sensitivity runs were made for the July 2010 episode using the 1995 meteorological data with a 2010 emission inventory. Sensitivity coefficients were computed each hour for all the grid cells in the domain. By examining sensitivities at a specific station, we can determine the sub-domain from which emission reductions would have the greatest effect. Figure 4 shows a 6-day stacked bar chart for daily averaged sulfate sensitivities at the Great Smokey Mountains National Park. The sulfate sensitivities represent the percent change in sulfate concentrations due to a 10% reduction in the west-inner sub-domain (WI), north-inner sub-domain (NI), south-inner sub-domain (SI), east-inner sub-domain (WI), all outer sub-domains (AO), and domain wide (sum of WI, NI, SI, EI, and AO). In the Great Smokey Mountains National Park, it can be seen that different sub-domains can have varying contributions to the overall reduction of sulfate from day-to-day depending on the specific meteorology. For example, on July 11, a 10% reduction in domain-wide SO2 emissions will result in a 3% reduction in the sulfate concentration at GSM, with 2% due to SO2 reductions in AO and 1% due to SO2 reductions in WI. It should be noted that the sensitivity results reported above are just an example of the type of results that are available. Since this is a work in progress and the data presented above is a partial data set, any inferences that might be drawn could be significantly altered when the rest of the data becomes available.
CONCLUSIONS
With the integration of the ISORROPIA aerosol module and the Reactive Scavenging Module into the URM model, a “one atmosphere” model has been created which can be used to predict aerosol concentrations and wet acid deposition mass fluxes for the SAMI modeling domain. Typically, the mean normalized error for most of the aerosol and wet deposition species is less than 50%. Through the use of sensitivity analysis, control strategies can be developed to effectively reduce the impact of aerosols and acid deposition on the southern Appalachian Mountain air quality.
ACKNOWLEDGMENTS
This project is funded by the Southern Appalachian Mountains Initiative. The authors thank the members of the SAMI Atmospheric Modeling Subcommittee for their constructive criticism, innovative ideas and invaluable help throughout this project.
REFERENCES
1. Berkowitz, C.E.; Easter, R.C; Scott, B.C. Atmos. Environ. 1989, 23, 1555-1571.
2. Carter, W.P.L. Atmos. Environ. 1990, 24, 481-518.
3. McNally, D.E.; Tesche, T.W. Modeling Analysis and Plotting System User Manual. Alpine
Geophysics, 1991.
4. Nenes, A.; Pilinis, C; Pandis, S.N. Aquatic Geochem. 1998, 4, 123-152.
5. Odman, M.T.; Russell, A.G. J. Geophys. Res. 1991a, 96, 7363-7370.
6. Odman, M.T.; Russell A.G. Atmos. Environ. 1991b, 25A, 2385-2394.
7. Odman, M.T.; Boylan, J.; Wilkinson, J.W.; Yang, Y.J.; Russell, A.G. this conference, 2000.
8. Pandis, S.N.; Harley, R.A.; Cass, G.R; Seinfeld, J.H. Atmos. Environ. 1992, 26A, 2269-2282.
9. Yang Y.J., Wilkinson J.G., and Russell A.G. "Fast, Direct Sensitivity Analysis of Multidimensional
Photochemical Models," Environ. Sci. Technol. 1997, 31, 2859-2868.
KEY WORDS
Air quality, aerosols, wet deposition, modeling, SAMI, model performance, sensitivity analysis